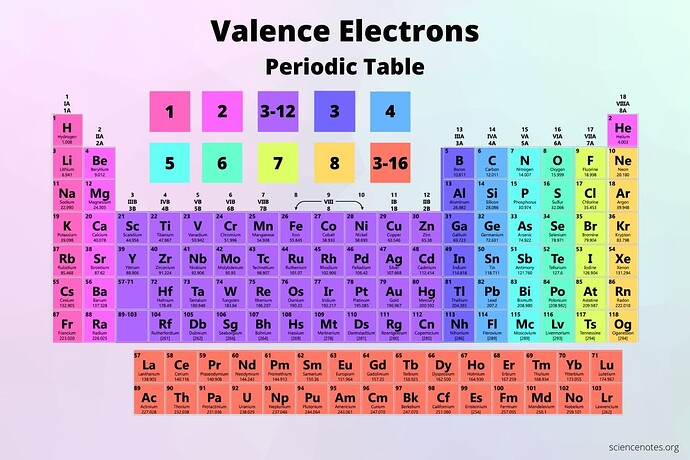
in semiconductors we are talking about groups of elements, but these groups repeat. what is the different between these groups ?
The group-4 elements are contained in the column labeled 4 with a blue color. Of those, C is a wide bandgap insulator (diamond), Si and Ge are semiconductors, and the rest are metals
thanks for the reply,
but how is the group 4 in the, 4rth column different ? (Ti, Zr, Hf, …)
There are many differences, such as the nr of electrons/protons, what shells are filled and what orbitals. Do you have some specific difference in mind?
i guess im just confused as to why theyre both called group 4 ? Since what we desire are elements with 4 valence electrons I suppose.
It just stood out to me the ‘‘group 4’’ wasent unique
so what do these ‘‘groups’’ represent if they repeat ?
indeed, calling both columns group 4 is confusing. But what is in a name? Some of them are indeed the 4th element in a row of the periodic table.
specifically the column group-4 (IVB) while the one we are considering is group-4 (IVA)