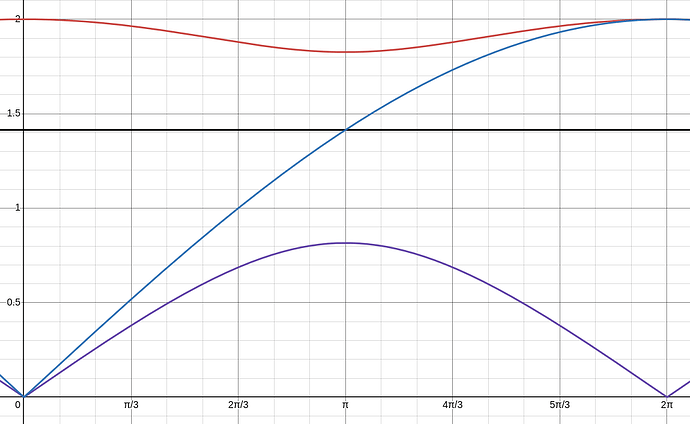
In this exercise, we have the dispersion relation
The answer states that the band gap is centered around \omega=\sqrt{\frac{\kappa}{\mu}}. I think this is not true. I suppose that in this case, to find the center of the band gap you can take the average of the upper band and lower band frequencies at ka=\pi. Generally, \sqrt{\frac{1}{2}(\omega_1^2 + \omega_2^2)} \ne \frac{1}{2}(\omega_1 + \omega_2) (unless \omega_1 = \omega_2), in other words, the average of omega is not the square root of the average of omega squared.
This can be seen when you plot the dispersion relation and \omega=\sqrt{\frac{\kappa}{\mu}}, the horizontal line is not exactly in the middle.